
Question Video: Calculating the Equilibrium Constant for Concentration Given the Initial Amount of Each Reactant | Nagwa

50 kg of nitrogen and 10 kg of hydrogen are mixed to produce ammonia. Calculate the ammonia formed and identify the limiting reagent in the production of ammonia in this situation.

stoichiometry - How to calculate the equilibrium constant for nitrogen, hydrogen, and ammonia? - Chemistry Stack Exchange
How to calculate the maximum mass of ammonia, NH3, that could be made from 42 tonnes of nitrogen and excess hydrogen - Quora

Calculate the pH of a 0.10 M ammonia solution . Calculate the pH after 50.0 mL of this solution is treated with 25.0 mL of 0.10 M HCl. The dissociation constant of ammonia, Kb=1.77xx10^(-5)

Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram

Question Video: Calculating 𝐾_𝑝 at Equilibrium for a Mixture of Nitrogen, Hydrogen, and Ammonia | Nagwa
How to calculate your Un-ionized ammonia levels Information below collected from: Francis-Floyd, Ruth, Craig Watson, Denise Pett

Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram


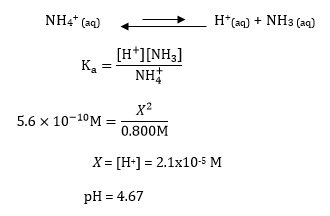






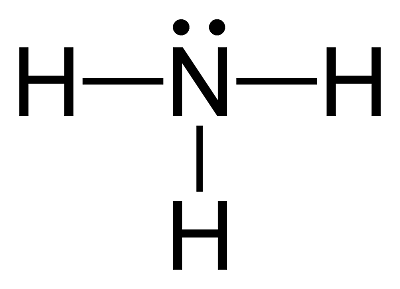


![Ammonia [NH3] Molecular Weight Calculation - Laboratory Notes Ammonia [NH3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonia-molecular-weight-calculation-300x150.jpg)




