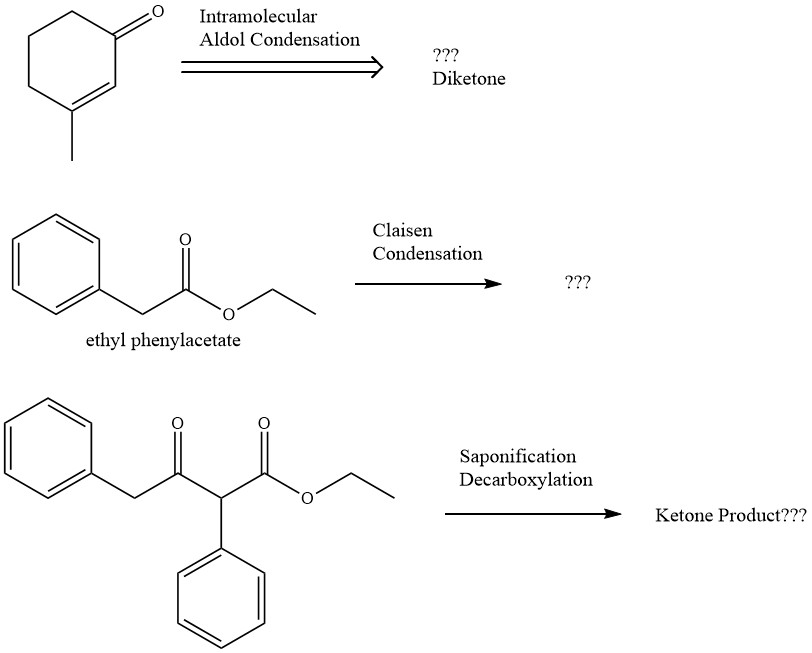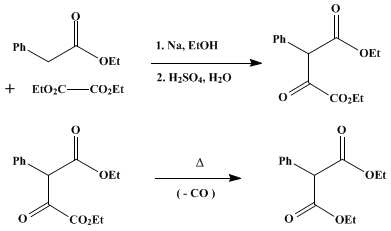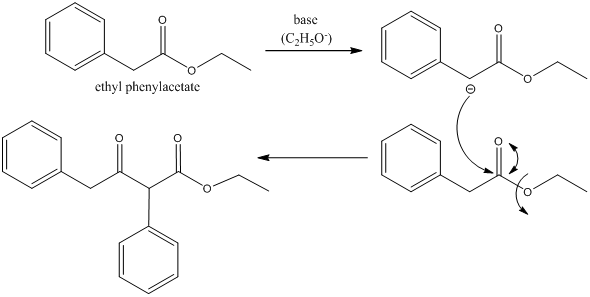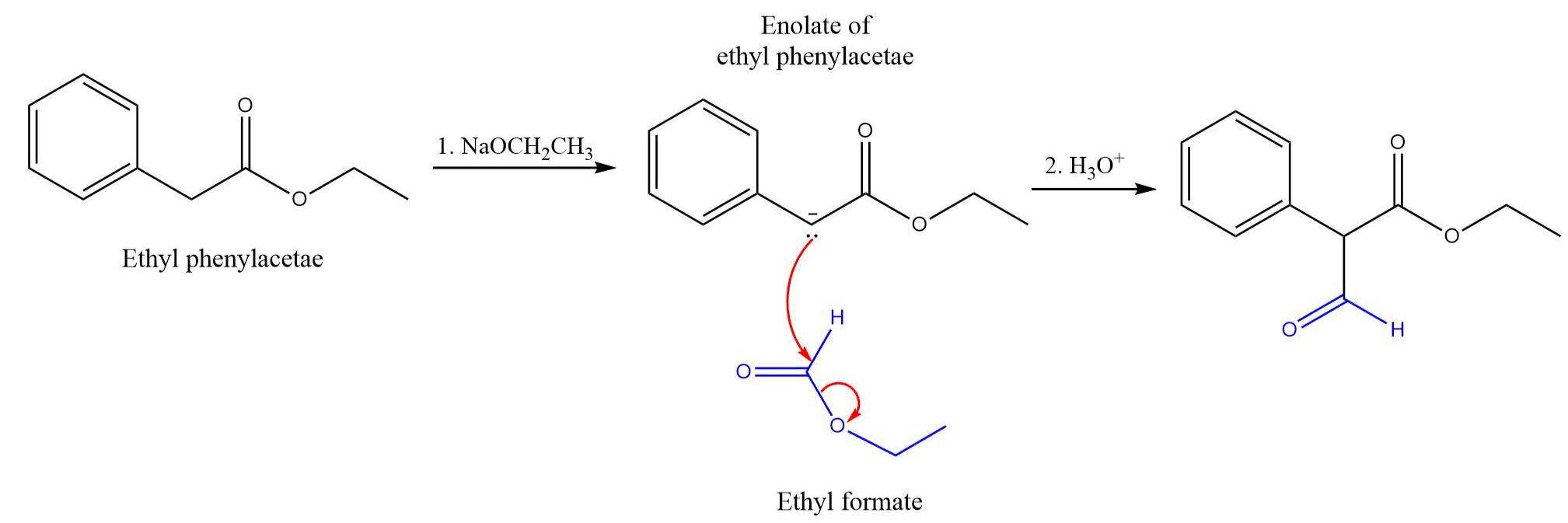
SOLVED:Show the products you would expect to obtain by Claisen condensation of the following esters: (a) (CH3)2 CHCH2 CO2 Et (b) Ethyl phenylacetate (c) Ethyl cyclohexylacetate

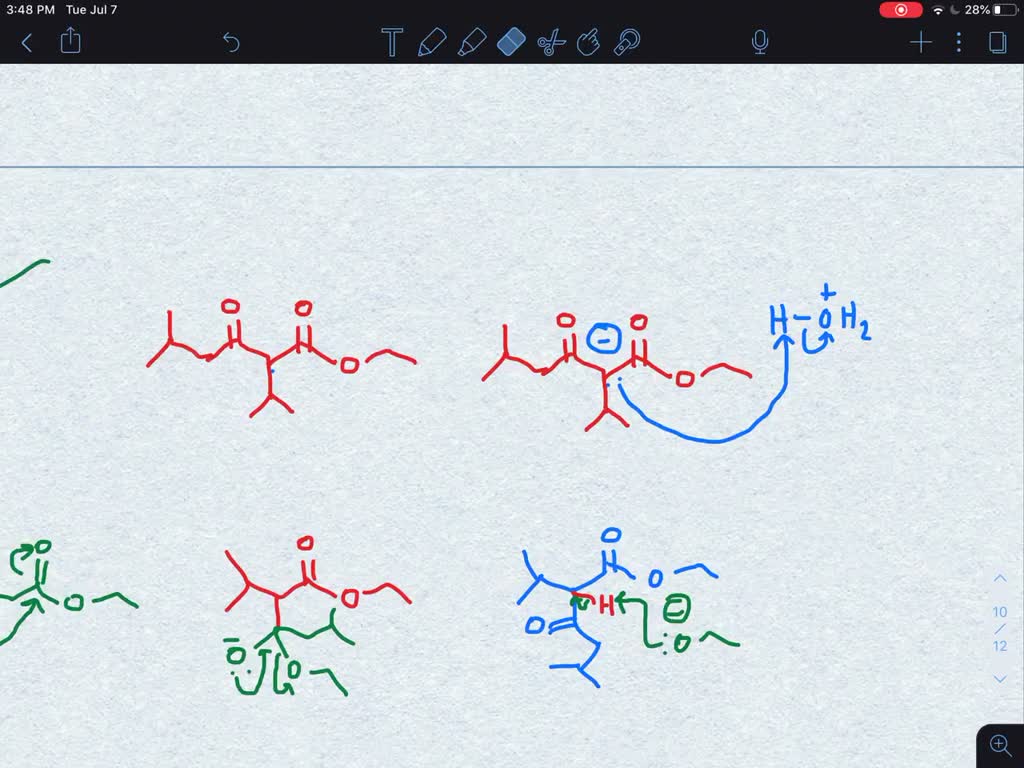
Draw the mechanism for the Claisen Condensation reaction below showing arrows and electrons movement. | Homework.Study.com

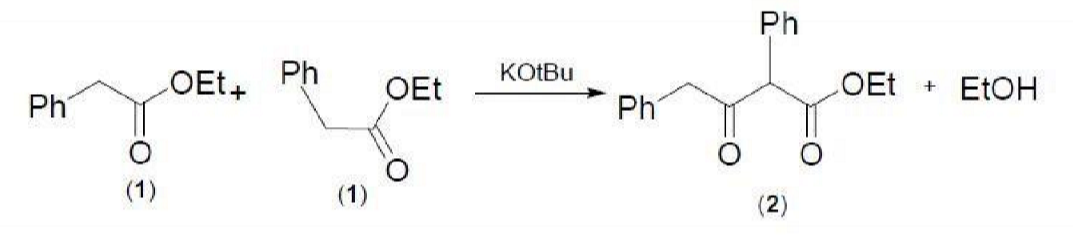
Draw the structure(s) of the product(s) of the Claisen condensation product of Ethyl phenylacetate (C6H5CH2COOCH2CH3). | Homework.Study.com

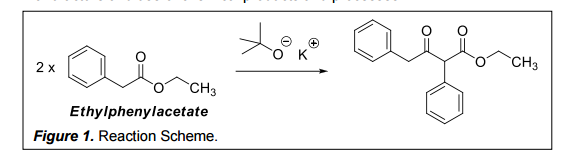
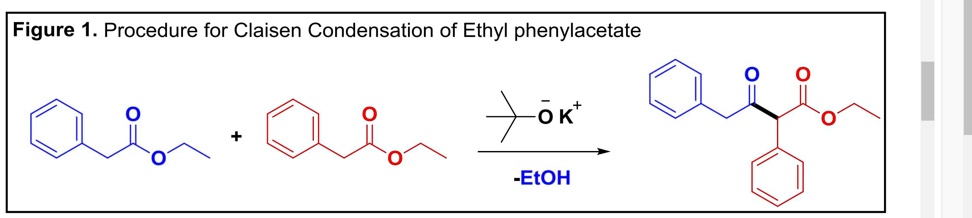
SOLVED: Experiment - Claisen Condensation Reactions: In this experiment, we performed a Claisen condensation reaction of ethyl phenylacetate using potassium tert-butoxide to form ethyl 3-oxo-2,4-diphenylbutanoate and ethanol without using a solvent. This
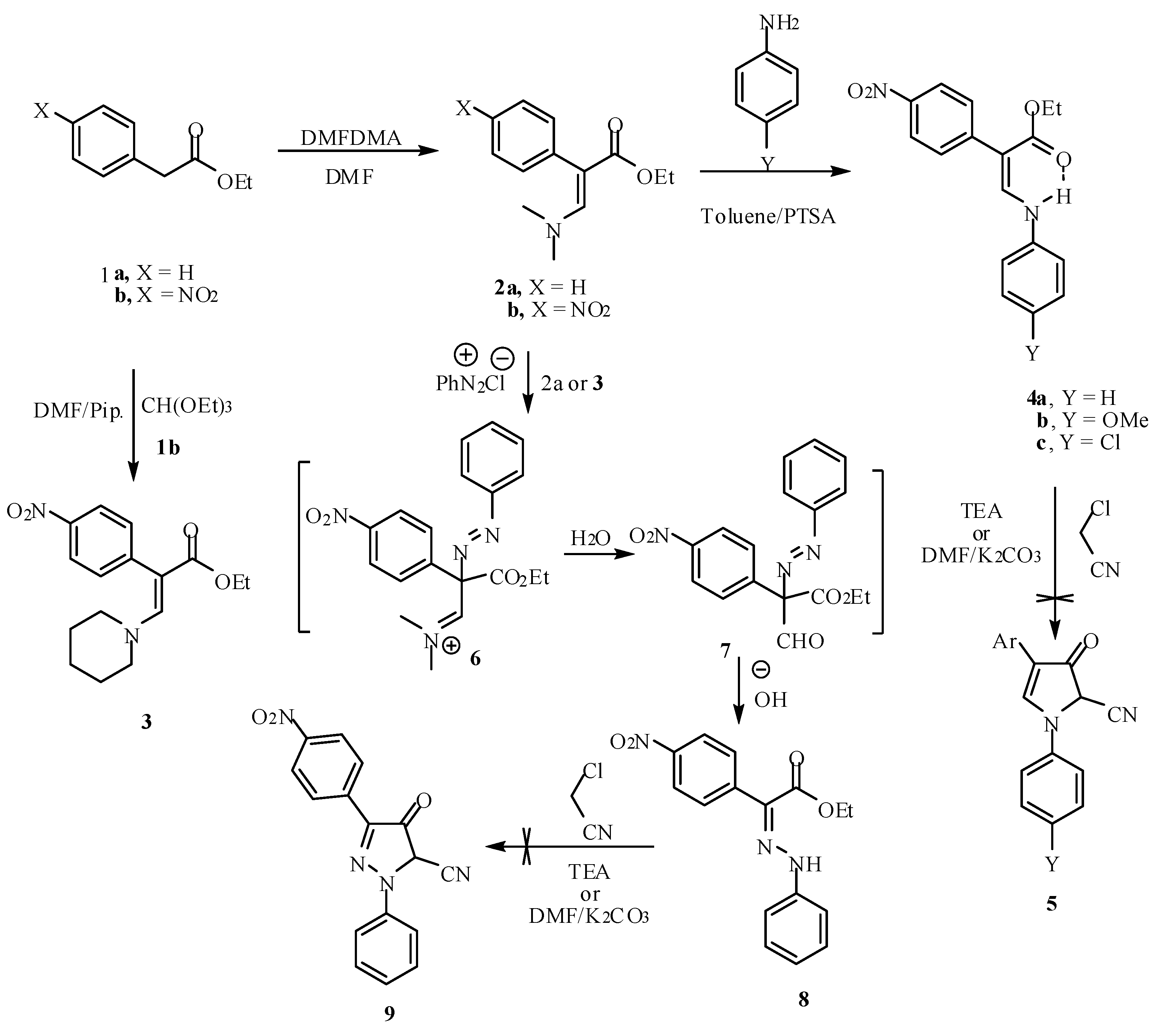
Molecules | Free Full-Text | β-Enamino Esters in Heterocyclic Synthesis: Synthesis of Pyrazolone and Pyridinone Derivatives