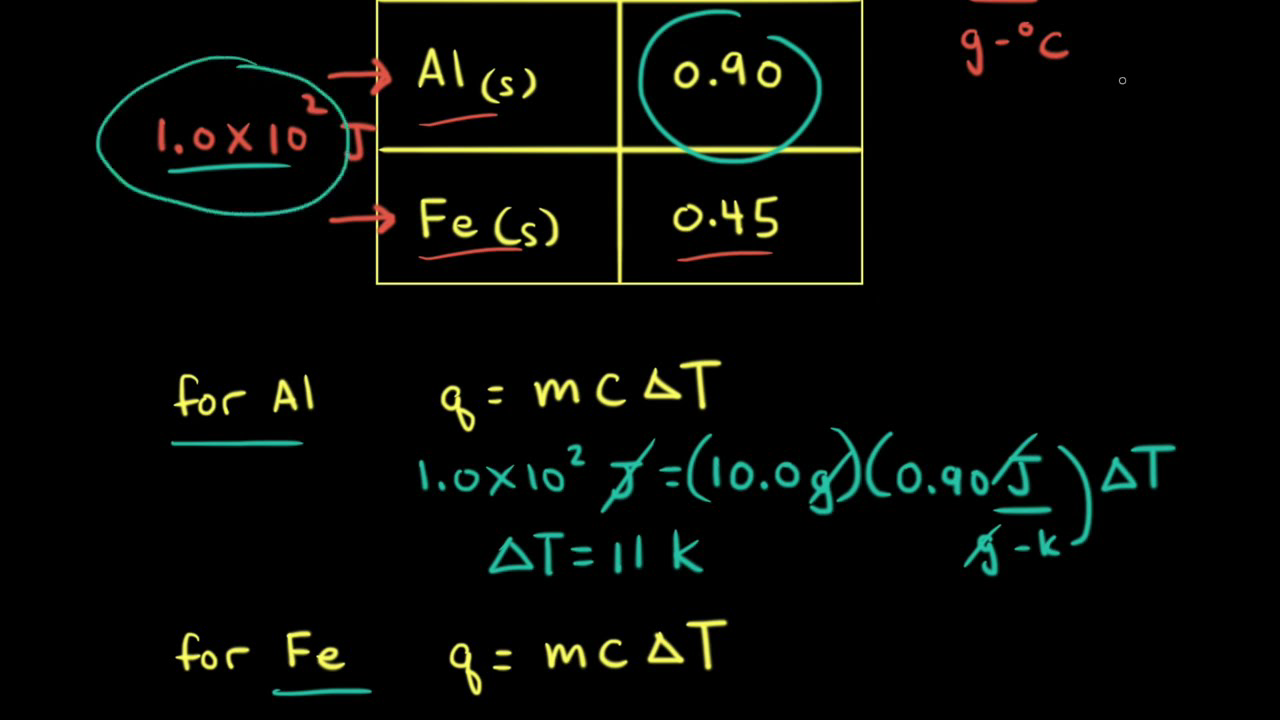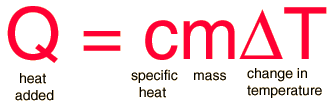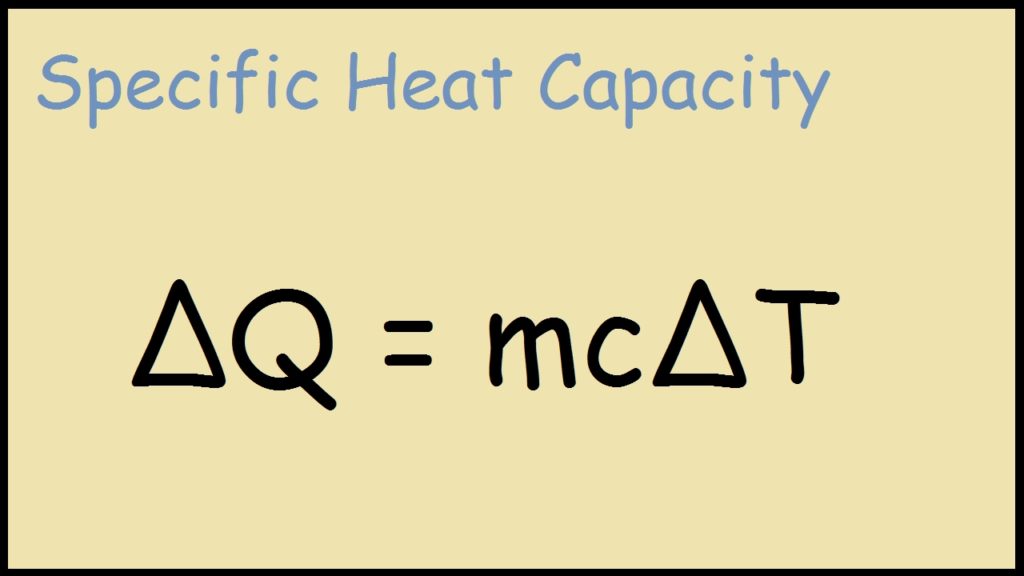SOLVED: Calculate the final temperature when 2400 joules of heat is given to an iron of mass 2kg at 20°C. (Specific heat capcity = 460 J/kg° C)
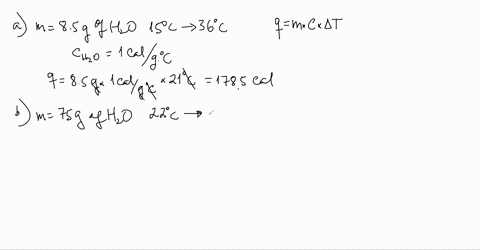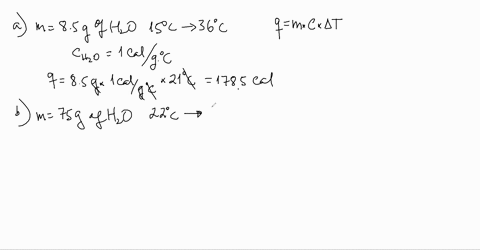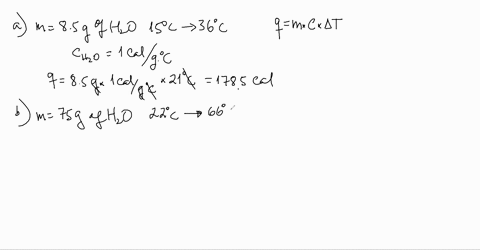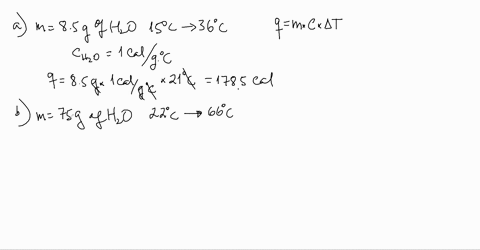
SOLVED:Use the heat equation to calculate the energy for each of the following (see Table 3.11): a. calories to heat 8.5 g of water from 15^∘ C to 36^∘ C b. joules

SOLVED: Calculate the heat capacity, in joules and in calories per degree, of the following: (a) 28.4 g of water (b) 1.00 oz of lead