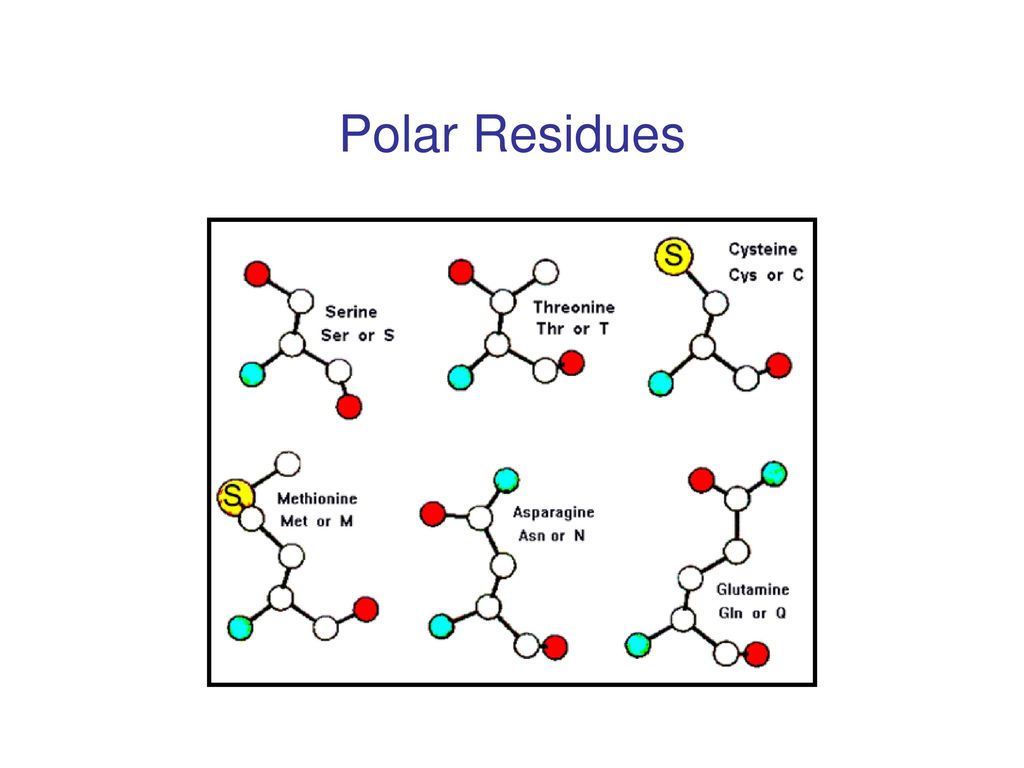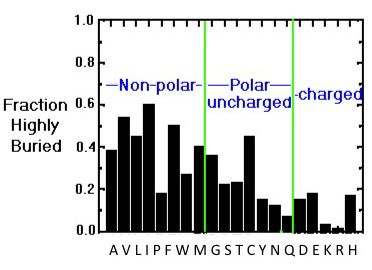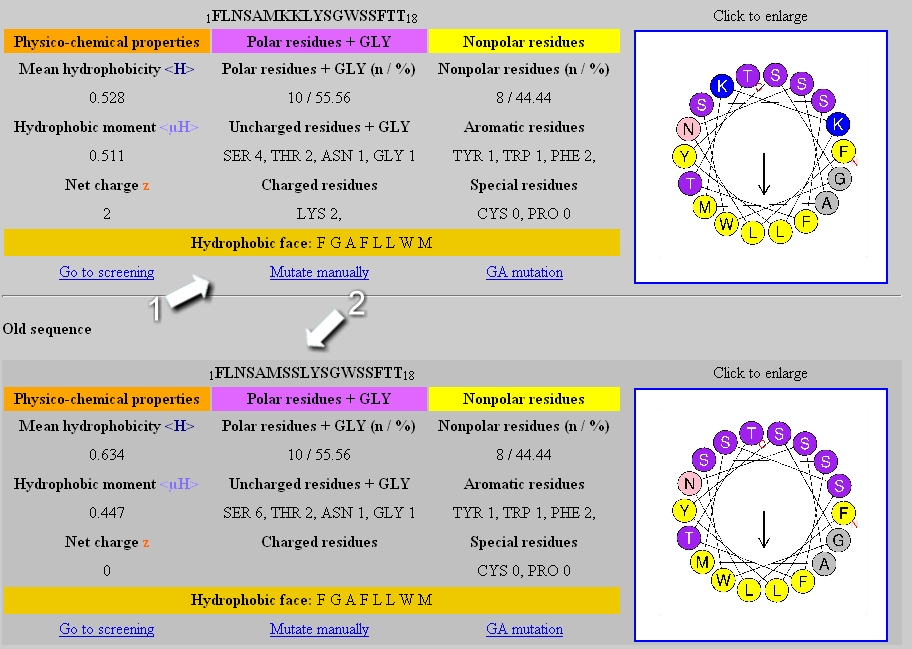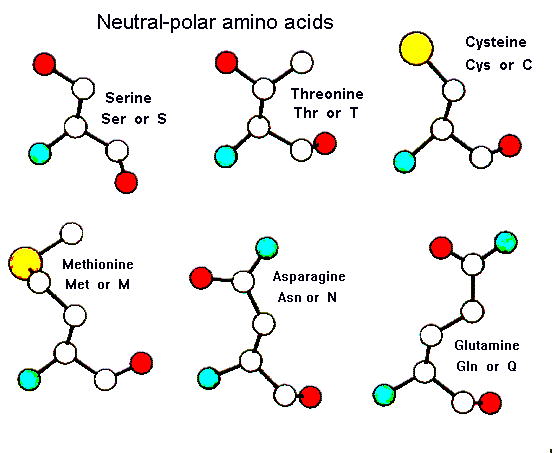
IJMS | Free Full-Text | Simple Models to Study Spectral Properties of Microbial and Animal Rhodopsins: Evaluation of the Electrostatic Effect of Charged and Polar Residues on the First Absorption Band Maxima

3D Interaction Homology: Hydropathic Analyses of the “π–Cation” and “π–π” Interaction Motifs in Phenylalanine, Tyrosine, and Tryptophan Residues | Journal of Chemical Information and Modeling

APH1 Polar Transmembrane Residues Regulate the Assembly and Activity of Presenilin Complexes - ScienceDirect

Why are polar residues within the membrane core evolutionary conserved? - Illergård - 2011 - Proteins: Structure, Function, and Bioinformatics - Wiley Online Library

Partitioning of hydrophobic and polar residues between the surface and... | Download Scientific Diagram

Propensities of Polar and Aromatic Amino Acids in Noncanonical Interactions: Nonbonded Contacts Analysis of Protein−Ligand Complexes in Crystal Structures | Journal of Medicinal Chemistry

Polar transmembrane interactions drive formation of ligand-specific and signal pathway-biased family B G protein-coupled receptor conformations | PNAS
Conserved polar/charged residues (in red; numbering as in MOR) that... | Download Scientific Diagram

How do non-polar residues (amino acid side chains) interact with other residues? How do polar residues interact with other residues? With water? How do charged residues interact with other residues? | Homework.Study.com

SOLVED: It depends on the occurrence of polar and nonpolar residues in sequence: Peptide #1 has every residue so it has more chance to be helical. On the other hand; peptide #2

Polar and apolar residues are segregated in AMPs to form amphilphilic... | Download Scientific Diagram
![PDF] On the significance of alternating patterns of polar and non-polar residues in beta-strands. | Semantic Scholar PDF] On the significance of alternating patterns of polar and non-polar residues in beta-strands. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/adc75e0f5e2d2d3c7ff74b6baa12a8882f1ff860/6-Table2-1.png)
PDF] On the significance of alternating patterns of polar and non-polar residues in beta-strands. | Semantic Scholar